|
Orbitals that overlap extensively form bonds that are stronger than those that have less overlap. The strength of a covalent bond depends on the extent of overlap of the orbitals involved. The mutual attraction between this negatively charged electron pair and the two atoms’ positively charged nuclei serves to physically link the two atoms through a force we define as a covalent bond. the single electrons in each orbital combine to form an electron pair.In a sigma bond, the electron pair occupies an orbital a region of space associated with a particular value of the energy of the systemlocated mainly between the. an orbital on one atom overlaps an orbital on a second atom and sigma bond, in chemistry, a mechanism by which two atoms are held together as the result of the forces operating between them and a pair of electrons regarded as shared by them.Evidence regarding the influence of caffeine and coffee on gastric emptying, appetite hormones, and appetite perceptions was equivocal. According to valence bond theory, a covalent bond results when two conditions are met: The literature review indicated that coffee administered 3-4.5 h before a meal had minimal influence on food and macronutrient intake, while caffeine ingested 0.5-4 h before a meal may suppress acute energy intake. We say that orbitals on two different atoms overlap when a portion of one orbital and a portion of a second orbital occupy the same region of space. 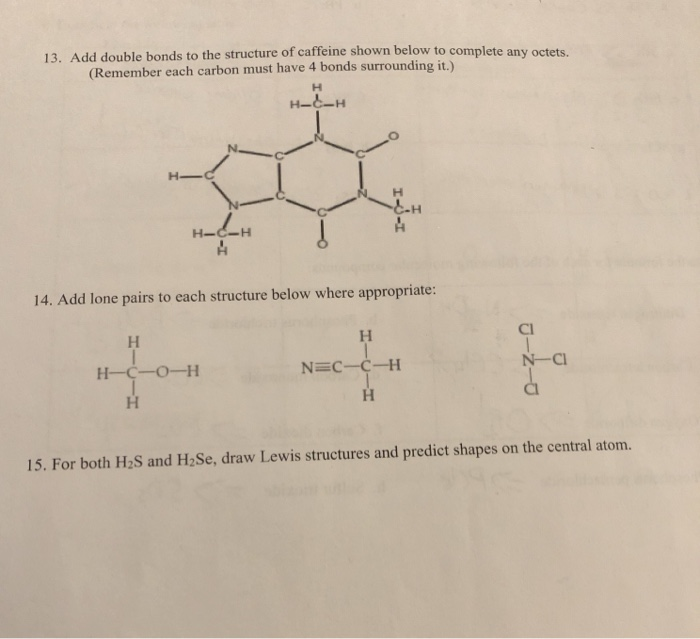
Valence bond theory describes a covalent bond as the overlap of half-filled atomic orbitals (each containing a single electron) that yield a pair of electrons shared between the two bonded atoms. Caffeine C8H10N4O - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety. \): Representative Bond Energies and Lengths Bond According to valence bond theory, a covalent bond results when two conditions are met: (1) an orbital on one atom overlaps an orbital on a second atom and (2) the single electrons in each orbital combine to form an electron pair.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
